|
8/11/2023 0 Comments Okazaki fragment sequencing Program in ensuring the inheritance of genetic information, the selection and activation of replication origins is remarkablyĭynamic and plastic. Despite the critical role of the DNA replication Origins across diverse cell types and developmental stages.Įvery cell cycle, tens of thousands of start sites of DNA replication must be established and coordinated to ensure the accurateĪnd complete duplication of >3 billion base pairs (bp) of DNA in the human genome. Primary sequence, chromatin environment, and nuclear architecture contribute to the dynamic selection and activation of replication Here we summarize recent advances in our understanding of how That define and regulate their distribution and utilization. Origins throughout mammalian genomes, providing an unprecedented opportunity to identify both genetic and epigenetic features The adaptation of origin-mapping assays to genome-wide approaches has led to the identification of tens of thousands of replication The limited number of identified origins prevented a comprehensive andĮxhaustive search for conserved genomic features that were capable of specifying origins of DNA replication. Of only a handful of mammalian replication origins. The development of molecular and biochemical approaches to identify start sites of DNA replication (origins)īased on the presence of defining and characteristic replication intermediates at specific loci led to the identification ).For more than three decades, investigators have sought to identify the precise locations where DNA replication initiates in.Additionally, the percent of the Okazaki fragment substrate converted to the RNA 12nt product in A was determined and also plotted ( The percent of acid soluble radioactivity (□) was determined for each fraction. RNase HI activity degrades the substrate releasing small oligonucleotides into the acid soluble fraction that were quantitated by scintillation counting. ( B) Random RNase HI cleavage was assayed on a poly(rA)/oligo(dT) substrate. Cleavage results in a specific RNA 12nt fragment and an oligonucleotide 39nt containing the remaining ribonucleotide and deoxynucleotides (indicated by arrows). Aliquots of the diluted fractions were assayed at 37☌ for 10 min. ( A) To generate a model Okazaki substrate, an internally radiolabeled ( 32P)RNA 13nt was extended with DNA 38nt and annealed to a DNA template. Aliquots (1 μl) of each fraction from the final column of the first preparation (Mono-Q) were diluted 1:4000 and tested for both RNase HI structure-specific and random cleavage activity.  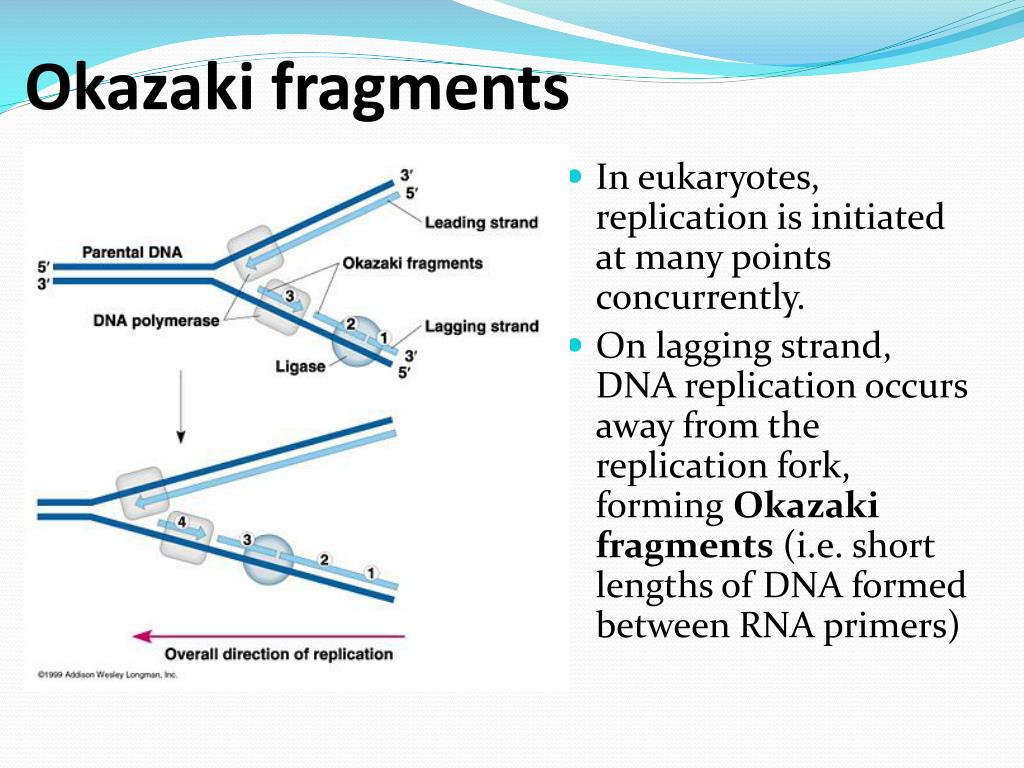
RNase HI contains both a structure-specific and random endonucleolytic activity. Because FEN1 prefers a substrate with a single-stranded 5'-flap structure, the single-stranded activity of junction RNase suggests that Okazaki fragments are displaced to form a 5'-tail prior to cleavage by both nucleases. After junction cleavage, FEN1 removes the remaining ribonucleotide. This activity exactly comigrates with RNase HI activity during purification strongly suggesting that both activities reside in the same enzyme. We have named this junction RNase activity. Clearly, the enzyme recognizes the transition from RNA to DNA on a single-stranded substrate and not the RNA/DNA heteroduplex structure. Furthermore, even fully unannealed Okazaki fragments were cleaved. In current work, highly purified calf RNase HI makes this exact cleavage in Okazaki fragments containing mismatches that distort the hybrid structure of the heteroduplex. Earlier evidence indicated that the cleavage site of RNase HI is 5' of the last ribonucleotide at the RNA-DNA junction on an Okazaki substrate. The initiator RNAs of mammalian Okazaki fragments are thought to be removed by RNase HI and the 5'-3' flap endonuclease (FEN1). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
